Carbonate rocks are important substrates for many vineyard soils. Limestone is the most common carbonate. It is a sedimentary rock that is widespread throughout the world. Dolostone or dolomite is the other significant carbonate rock. Combined, they cover around 15% of the world’s land surface. Limestone is based on the mineral Calcite, or Calcium Carbonate. In its simplest form, it is a calcium ion (Ca++) bonded with a carbonate ion (CO32- ) to form calcium carbonate (CaCO3). Ions other than calcium can combine with carbonate. In the case of dolomite, magnesium replaces some of the calcium and the result is [CaMg(CO3)2]. Other elements occasionally enter the equation, but calcium is the most important element for our concern. Calcium carbonate is soluble in weak acids and carbonate terrains are most noted outside the wine world for their propensity to form large caves as well as house-swallowing sinkholes.

Cliffs of Dolomite in the Alto Adige
Limestone soils get a great deal of adulation from students of wine. Many of the great winegrowing areas of France are underlain by limestone terroir. Chablis, the Côte d’Or, Pouilly-Fuissé in the Mâconnais, much of St.-Émilion, Champagne, Sancerre in the Loire Valley, and many of the Grands Crus of Alsace all have limestone as a base for their soils. While it is tempting to presume that limestone, by definition, makes the best vineyard soils, does it yield magnificent wines wherever it is found? Let’s investigate the particulars of this widespread rock type.
While they share a name, all limestones, are not the same. The term limestone refers to rocks composed of 50% or more of calcium carbonate. That means 49% of the rock can be something else – clay, sand, silt, gravel or whatever. If a Pinot Noir were blended with 49% Syrah, it’s likely that the Syrah would dominate the blend. So too, limestone can be dominated by the characteristics of its non-carbonate components. Thus, we have a complete spectrum of rocks – a calcareous clay with 49% CaCO3 becomes an argillaceous limestone at 50% CaCO3. As scientific and precise as the geologic terminology sounds, it is far from a definitive statement.
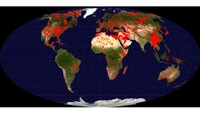
Worldwide Distribution of Carbonate Rocks (in red)
Limestone forms in warm, shallow seas. Modern limestones are being created in the Bahamas and in coral reefs dotting tropical waters around the world. Most limestones are composed of accumulated fragments of organisms that secrete calcium carbonate in their shells. Seawater has high concentrations of dissolved calcium and bicarbonate ions, so corals, clams, oysters and other sea creatures have developed the ability to synthesize these ions into hard, calcium carbonate shells. Because they contain numerous fossils, geologists study carbonate rocks closely. The fossil evidence enables a reconstruction of the depositional environment and an ability to correlate with geologic formations in other parts of the world. Dolomite is thought to form when magnesium-rich seawater percolates through existing limestones, replacing some of the calcium ions with magnesium.
The ancient sea dwellers that form limestones around the world can be large and visible as fossil shells a meter long or tiny, with their calcareous shells visible only under the microscope. The famous chalk of Champagne is composed of the skeletal remains of billions of tiny algal organisms called coccoliths. The famous Kimmeridgian clay/limestone of Chablis and beyond is noted for its prolific fossil content.

Kimmeridgian Age Fossil
Rock deposition is hardly a sanitary process, so sands, gravels and clays from other sources can be mixed in with the shell fragments resulting in sandy limestones, limy clays, and sandy-gravelly-limestones in all permutations. Other minerals like hematite or glauconite can get into the mix as well, resulting in red or green tints, respectively, to the rocks.
What is special about limestone then, that makes it such a mythical vineyard soil? Clearly, there are vines that benefit from limestone soils. One needs only to point to Chablis for a stellar example of “limestone minerality.” The best Chablis wines, however, are grown on Kimmeridgian sediments, which are as much clay as they are limestone. Which is the most important component? Perhaps both are needed to accomplish the delicious miracle of Chablis. The great Grands Crus of the Côte de Nuits are grown on marl – a mixture of soft limestone and clay. It’s likely that the combination makes the soil especially beneficial to Pinot Noir vines.
Limestone normally evolves into a higher pH soil. There is an observable inverse relationship between soil pH and the pH of resultant wines. Chardonnay grown on high pH soil (e.g. Chablis) yields lower pH wines. In other words, higher acid wines. Research suggests as well that nutrient uptake at the vine rootlet is greater when the pH is moderate (a 6-7 level). Calcium ions, along with magnesium, potassium and sodium ions (all cations in the chemical parlance) can be transmitted across the rootlet cell walls when the pH and water presence are ideal. The enhanced cation exchange capacity or CEC is one of several measurements that help define optimal soils for viticulture. Higher acid (lower pH) soils provide a less favorable transfer of ions. Adding powdered limestone to these low pH soils has proven beneficial for plant growth – a testament to the power of the rock.
Too much carbonate, however, can be detrimental to vines. The vine malady chlorosis is often related to excessive lime and too high a pH in the soil. Chlorosis is a yellowing of plant leaves due to an iron deficiency. The lack of green matter results in weak photosynthesis and weak plant vigor. High lime soils cause iron to react and be bound up in insoluble forms and unavailable to the plant. This became particularly evident in post-phylloxera times, since North American rootstocks did not take well to the high lime content of many French soils. Obviously, too much limestone is a hindrance to great wines and, once more, we must seek balance among components to achieve greatness.

Characteristic Yellowing of Chlorosis-Affected Vine Leaves
As we have noted, limestone is praised as a vineyard soil in many parts of France. Much is made of the few areas of limestone soils in California – Chalone, Mt. Harlan and a few others – as superior sites for Pinot Noir and Chardonnay. Limestone provides a good habitat for many other grape varieties around the world. The higher elevations of Tuscany are noted for limestone soils and Sangiovese wines. South Australia’s Coonawarra is noted for both its ‘terra rossa’ limestone soil and its Cabernet Sauvignon wines. The Right Bank of Bordeaux has abundant limestone in the soil and grows primarily Cabernet Franc and Merlot. Limestone underlies vineyards of Negroamaro grapes in Apulia, and Verdicchio grapes in Marche. There are large areas of limestone-based soils in Greece. Carbonate rocks underlie the Agioritiko grapes in Greece’s Nemea region as well as the Xinomavro grapes in Naoussa and Amynteon.

Terra Rossa Limestone Soil in South Australia
It is clearly evident that all of the grape varieties grown successfully in carbonate-based soils can also produce fine wines in areas with no limestone. The Pinot Noirs of Oregon are grown successfully on siltstone and basalt-based soils. Is there a common thread of greater finesse and complexity among carbonate-based wines? That’s an argument that will likely never be solved but will be fun to pursue. To sum up, carbonate rocks are just a piece of the puzzle in the grand complex of factors that produce the amazing diversity of the wines we love.